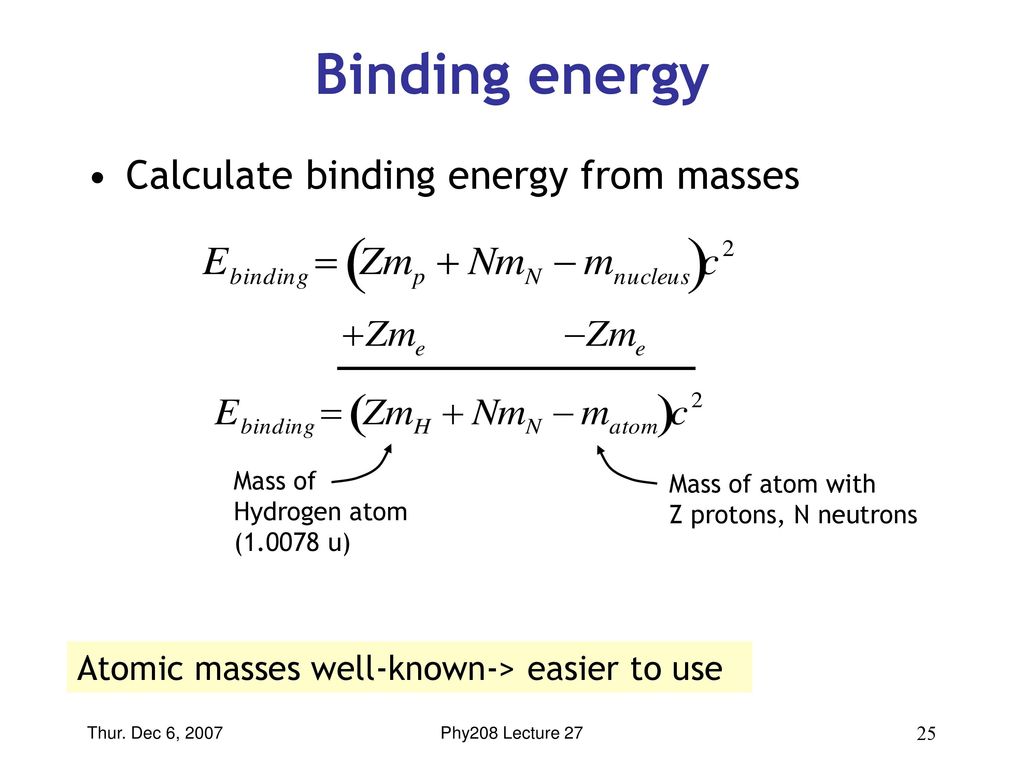
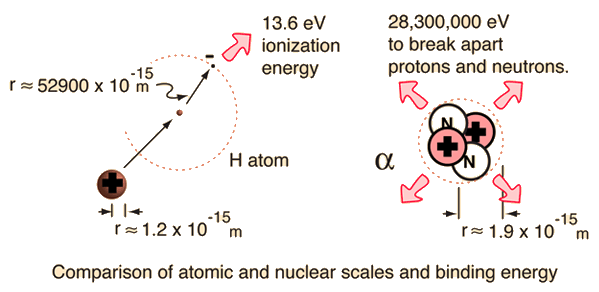
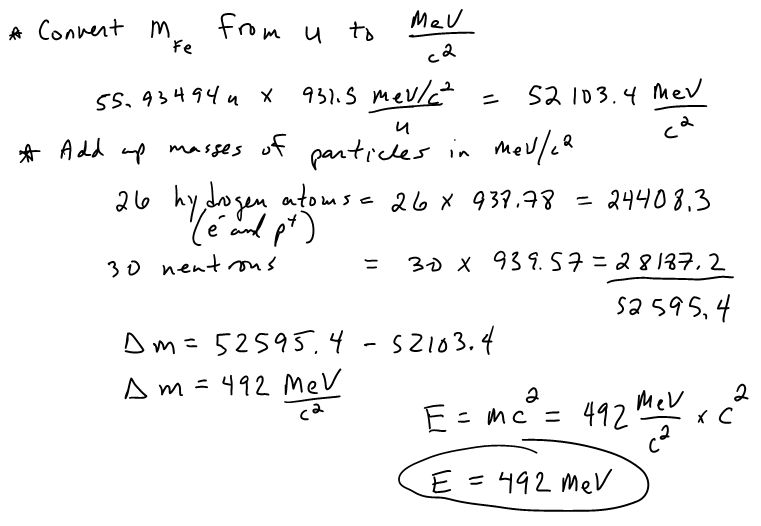Relationship between hydrogen binding energy and activity for hydrogen evolution reaction by palladium supported on sulfur-doped ordered mesoporous carbon - ScienceDirect

If the binding energy of 2nd excited state of hydrogen like sample is 24ev approx,then determine the atomic number of - Chemistry - Structure of Atom - 12877143 | Meritnation.com

In a hydrogen atom, the binding energy of the electron in the nth state is En ,then the frequency of revolution of the electron in the nth orbit is:
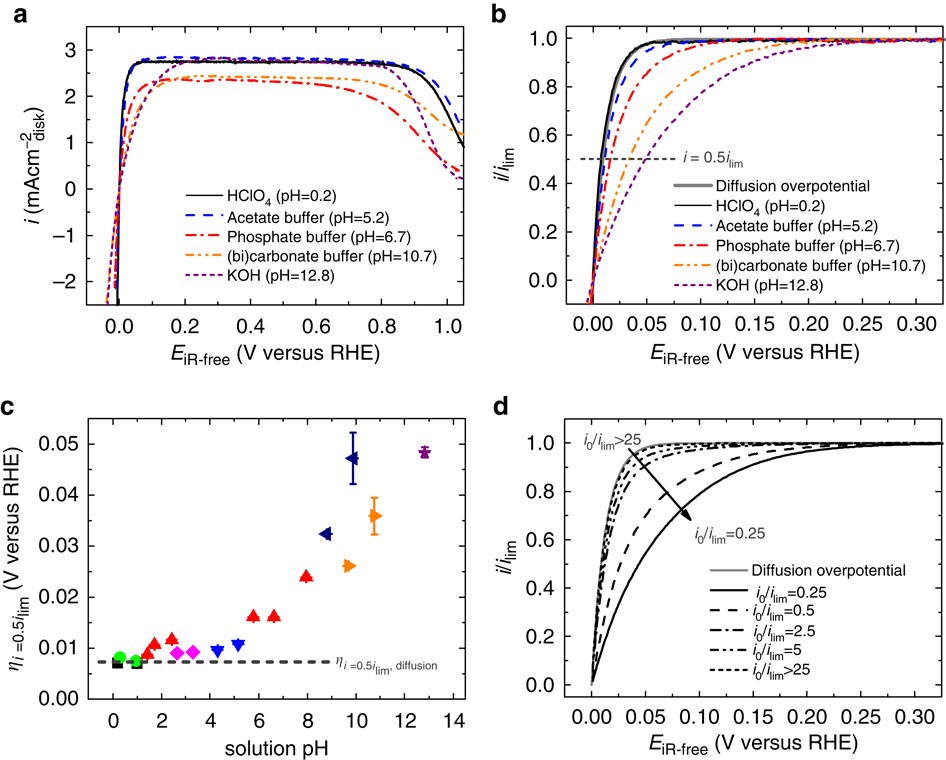
Correlating hydrogen oxidation and evolution activity on platinum at different pH with measured hydrogen binding energy | Nature Communications

Find the binding energy of the electron in the ground state of hydrogen-like ions in whose - YouTube
If the binding energy of the electron in the ground state of hydrogen atom is E, then the frequency of electron in the nth orbit is

The binding energy of e^(-) in ground state of hydrogen atom is 13.6 eV . The energies required to eject out an electron from three lowest states of He^(+) atom will be (

The binding energy for the nth hydrogen atom by the most stable vac +... | Download Scientific Diagram

In a hydrogen atom, the binding energy of the electron in the ground state is `E_(1)` then - YouTube

A hydrogen atom in a state having a binding energy of 0.85 eV makes transition to a state with - YouTube

Hydrogen nucleus combines to form helium then energy is released. Binding energy/nucleon of `He` is - YouTube

If the binding energy of the electron in a hydrogen atom is 13.6 eV, the energy required to remove the electron from the first excited state of Li^(++) is
If the binding energy of the electron in a hydrogen atom is 13.6 eV, - Sarthaks eConnect | Largest Online Education Community